
August 2019
Diagnosing Intestinal Parasites of Dogs and Cats
By Dr. Brian Herrin
Like all skills, becoming more comfortable diagnosing parasites in cats and dogs takes practice. We’ve previously discussed how to run a high-quality fecal floatation, but in this article we’ll talk more about how to interpret those tests.
Preparing the team
It can take some time for vet nurses or assistants to become proficient in diagnosing the common parasites of dogs and cats. Having a good reference nearby can help with that process. Zajac and Conboy’s “Veterinary Clinical Parasitology” is available for purchase as a lab manual or digital copy. It provides instructions on how to run a variety of techniques, as well as color pictures of the most common parasites found in the major domestic species. (All profits from the book also go to fund student travel grants to the annual American Association of Veterinary Parasitologist’s meeting.) Another good resource for color images is the Companion Animal Parasite Council’s website (www.capcvet.org).
It is also helpful to have a positive control sample for training or comparison purposes. If you have a fecal sample that is LOADED with parasites, add 10% formalin at a 1:4 formalin:feces ratio and place the sample in the fridge. The sample will remain diagnostic for at least a year if not longer. You can mix samples of multiple different parasites into one. Our lab has a hookworm/roundworm/ whipworm/coccidia sample that we keep for training and teaching purposes. When training new staff, we will show them the parasite egg of interest and ask them to find more on the slide. The next day we will repeat the same, allowing them to find the parasite on their own and recognize the inherent variation between eggs. Then on the third day, the technician will be asked to find the parasite on their own, and most students/technicians are very capable at that point.
The rest of this article will cover some of the major features of the eggs/oocyts/cysts that are commonly found in fecal floats of dogs and cats.
Roundworms - Toxocara canis and Toxocara cati
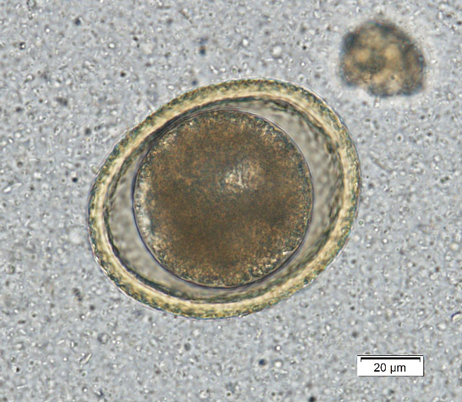
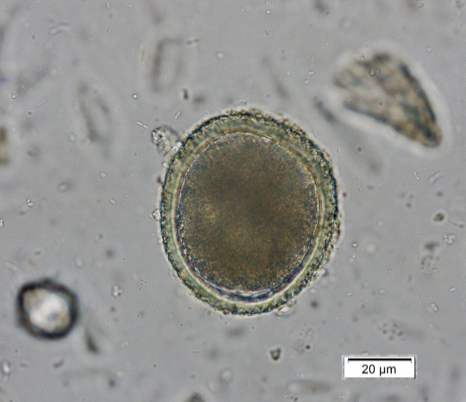
Toxocara canis is the dog roundworm and T. cati is the cat roundworm. Their species’ names help keep them straight, and there are some morphologic characteristics that help us differentiate them under the scope. Both T. canis (Figure 1a) and T. cati (Figure 1b) are round to oval eggs with thick, pitted, golden yellow shells. Within the shell is a dark brown developing embryo that is only one circular mass in fresh samples but can start to divide as a sample ages. Toxocara canis is approximately 75 µm x 85 – 90 µm compared to the 65 µm x 75 µm of T. cati, which make size the primary differentiator between these two eggs.
In addition, the pitting of the shell of T. canis makes the egg look like a golf ball with dimples when you focus up and down, while T. cati has more pin prick-like pitting. Finally, the developing embryo of T. cati fills the shell almost completely, while T. canis has some extra space within the shell. The appearance characteristics can be variable, and therefore egg size should be the main characteristic used for interpretation.This emphasizes the point of having a micrometer eyepiece for your microscope. These can be purchased for all common microscopes for approximately $50 online.
Think this doesn’t matter? A study in Switzerland found that 31.5% of roundworm eggs in dog fecals were T. cati! That means the dogs were NOT infected but had been eating cat feces!
|
|
| Figure 1a) Toxocara canis egg. Note the extra space around the embryo and the dimpled pitting of the shell. | Figure 1b) Toxocara cati egg. Note that it is smaller than T. canis and does not have extra space within the shell. |
Hookworms - Ancylostoma caninum
For hookworms, this article will focus on Ancylostoma caninum in dogs. While A. braziliense can infect dogs and cats, it is most commonly found in southern Florida, the Caribbean, or South America. If your patient has traveled to those areas, then you may find A. braziliense on the flotation, but luckily the eggs are morphologically similar and are treated the same as well.
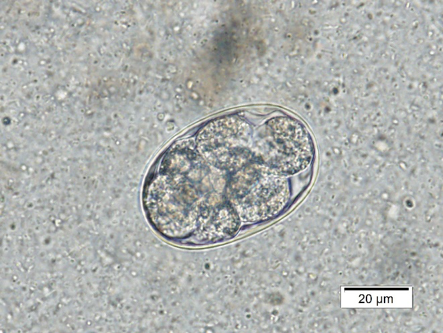
The eggs of A. caninum are an elongated oval approximately 50 – 70 µm in length by 30 – 50 µm in width. The egg has a smooth, thin shell with a dark gray-brown developing embryo. The embryo can be at a variety of stages of development and sometimes looks like one mass within the cells, while other times 8-16 distinct, round cells can be visualized (Figure 2a).
Hookworm eggs must be differentiated from strongyle-type eggs (Figure 2b) found in ruminant and equine feces. Most hookworm eggs will be <60 µm in length, while the other strongyle type eggs are >60 µm. If coprophagy is suspected, the patient can be kept away from the source (i.e. pasture, barn, etc.) for two days and another fecal float performed. If the eggs are strongyle type eggs from coprophagy, the fecal will be negative at that time.
 |
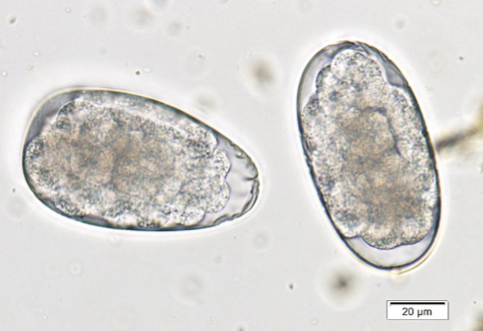 |
| Figure 2a) Ancylostoma caninum. Not the circular, defined cells of the developing embryo. | Figure 2b) Strongyle-type eggs from a horse. These eggs can be mistaken for hookworms but are longer than the normal hookworm eggs. Note the size bars in the right corner of each picture. |
Don’t miss Part 2 of Dr. Herrin’s article in the next issue of Diagnostic Insights for Veterinary Nurses. He covers whipworms, giardia and coccidia! And always remember, the parasitology service of the Kansas State Veterinary Diagnostic Laboratory is available to assist you with any challenging cases you encounter.
Next: New Test Offerings
Return to Index