The Wizz Quiz: Understanding Urinalysis
By Brandy Kastl, DVM
Importance of a Urinalysis:
A urinalysis provides information about the overall health status of our patients. It is easy to remember that diseases in the kidneys and urinary bladder can be detected on a urinalysis. But diseases in the liver, pancreas, heart, blood vessels, adrenal glands or brain may also cause abnormal urinalysis results. A properly collected and performed urinalysis is a recommended part of the minimum diagnostic work-up. Reaching a correct diagnosis in our patients requires not only an understanding of animal diseases, but also how urinalysis results can be affected by sample handling and collection methods. In some instances, results are so severely affected that an accurate diagnosis cannot be made.
Sample Collection:
|
|
|
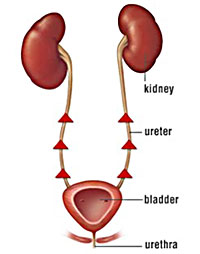
| Figure 1. Vesicoureteral Reflux |
Preferred sample collection methods depend on the suspected disease process, patient species and temperament, technical skills of the collector, and available technology or resources (See Table 1.) Regardless of method used, always collect as much urine as possible, preferrably at least 5 mL. Sample collection methods can sometimes affect urinalysis results. For instance, urine collected from voided samples has the highest risk of bacterial contamination. Often, voided urine samples are collected from floors, exam tables, reused collection containers (ladles, pie tins, cups, etc..), or litter pans which can all contain bacteria. Also, voided urine samples may pick up bacteria from the skin or fur of veterinary patients increasing the risk of contamination. In cases of suspected urinary bladder infections or high urine protein concentrations in small animals, samples collected via cystocentesis are best. Bacteria present in cystocentesis samples represent bacteria in the urinary bladder (i.e. bladder infection). When a cystocentesis cannot be performed, sterile urinary catheterization is recommended. Manual bladder expression is not recommended as a routine urine collection method due to vesicoureteral reflux (Figure 1). When a full urinary bladder is squeezed, urine will flow up the ureters toward the kidneys before exiting the body via the urethra. If a urinary bladder infection is present, bacteria is pushed up into the kidneys and could result in a severe kidney infection (i.e. pyelonephritis).
|
Method |
Pros |
Cons |
|
Voiding |
Easy Little patient risk |
High risk of bacterial contamination Uncooperative patients |
|
Catheterization |
Less risk of bacterial contamination Relatively safe |
Technical skill needed May need sedation |
|
Cystocentesis |
Best sample type for a bladder infection Relatiely safe Often no sedation needed |
Technical skill needed Blood contamination common Risk of bladder rupture or bleeding |
|
Manual Expression |
Not recommended due to vesicoureteral reflux (Figure 1) |
High risk of pushing bacteria from the bladder up to the kidneys |
Table 1. Comparison of Urine Collection Methods
3 T’s of Urine Sample Handling:
- Tubes: A new sterile container should be used for every sample.If a urinalysis will be completed in house, the sample may be placed into a conical urine sediment tube immediately after collection ( estimated cost/tube ~$0.03).In the author’s experience, pouring off unwanted urine after centrifugation is easier with a conical tube than a red top tube (estimated cost/tube ~$0.10).Alternatively, urine should be placed in a red top tube and stored immediately in the refrigerator.If a urine culture is wanted, remember to remove needles from syringes and tops from tubes before putting in the urine in order to prevent accidental bacterial contamination.
- Time: Completing a urinalysis within 1 hour of sample collection will give the most accurate results.If testing cannot be completed within 1 hour, samples should be placed in a red top tube and refrigerated.Any bacteria or cells present in a urine sample kept at room-temperature for more than 1 hour will continue to use glucose (sugar) in the urine.This may result in a falsely decreased urine glucose measurement.Also, bacteria produce ammonia which makes the urine more alkaline (increases pH).Finally, this increase in urine pH causes cells, like white blood cells, to degrade and may become unrecognizable.
-
Temperature: Every urinalysis should be performed on room-temperature urine. If urine was refrigerated, allow the sample to sit at room temperature for 30 minutes before performing the urinalysis.Cold urine can cause a false increase in the urine specific gravity measurement potentially resulting in an incorrect diagnosis in our patients.Refrigerated urine samples, even those allowed to return to room temperature, may have amorphous crystals on the urine sediment (Figure 2).These crystals are a temperature artifact and do not inidcate disease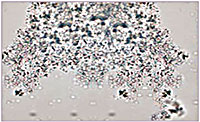
Figure 2. Amorphous Crystals in Urine
Things to Remember:
- Urinalysis results give information not only about the kidneys, but about the body as a whole.
- Urine collection preference order: cystocentesis > sterile urinary catheterization > clean void > contaminated void
- In-house urinalysis should be completed within 1 hour.Otherwise, refrigerate until ready.
- Allow urine sample to return to room-temperature before performing urine specific gravity.
- Dip the urine dipstick.
- Try an air-dried, Diff-Quik® stained urine sediment smear to see cells and bacteria better.