Canine Bacterial Pyoderma: Indications and Skin Sampling Techniques
By Dr. Chanran Ganta and Dr. Mary Bagladi-Swanson
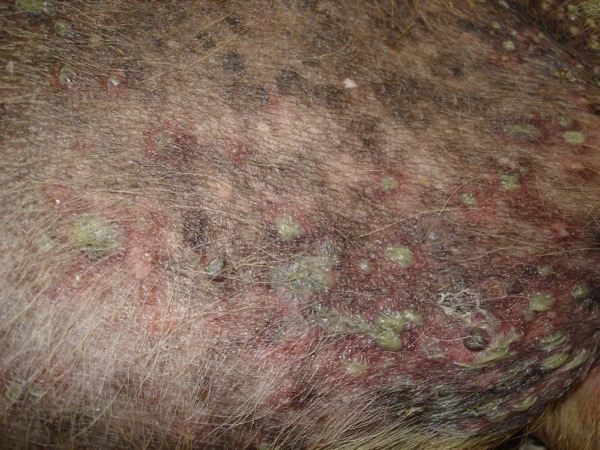
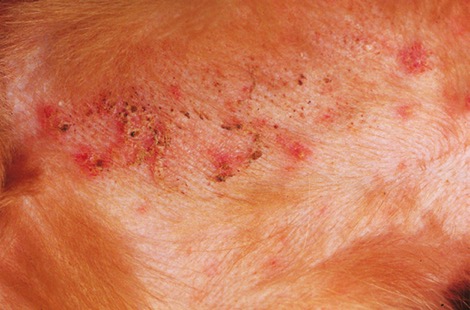
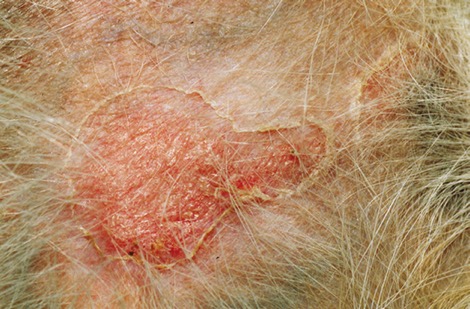
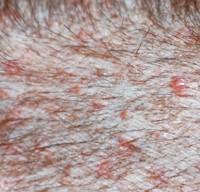
Canine bacterial pyoderma is a common skin condition that often has a prolonged course of infection, posing a significant challenge for disease treatment and management.
Staphylococcus pseudintermedius is the most common organism associated with canine pyoderma. Normal commensal bacteria like Pseudomonas, Enterobacter, E. coli, etc. can transform into primary pathogens due to abnormal ‘host factors’ such as hypersensitivities, ectoparasitism, endocrinopathies, autoimmune diseases and cornification abnormalities.
Successful treatment of canine bacterial pyoderma requires identifying the underlying skin condition, isolating the bacterial pathogen through culture, and identifying its antimicrobial susceptibility. The procedures to collect samples for culture and susceptibility depend on the lesion type and are described in Table 1.
Indications that suggest bacterial culture may be required in canine pyoderma cases
- Presence of intracellular rod-shaped bacteria on cytology
- Previous history of drug-resistant infection in the dog or in a pet from the same household
- Less than 50% reduction in clinical improvement within 2 weeks after appropriate systemic antimicrobial therapy has begun
- Emergence of new lesions (papules, pustules, collarettes) 2 weeks or more after the initiation of appropriate antimicrobial therapy
Table 1. Sampling guidelines for lesions of superficial bacterial pyoderma for culture and susceptibility testing
|
Lesion |
Sampling Procedure |
|
Pustule
|
No surface disinfection. Clip hair with sterile scissors (avoid clippers). Lance pustule with sterile narrow-gauge needle. If purulent exudate is visible on the needle, apply to a sterile swab; if not, gently touch exudate expelled from pustule with sterile swab and place in transport medium or sterile container. Sometimes lancing of very small pustules results in haemopurulent exudate, which is still suitable for sampling. |
|
Crust
|
No surface disinfection. Use sterile forceps or a sterile needle to lift the edge of a crust. The presence of exudate under a crust indicates an ideal site for culture. Touch sterile swab to exposed skin surface and place in transport medium or sterile container |
|
Epidermal collarette
|
No surface disinfection. Clip hair with sterile scissors (avoid clippers). Roll sterile swab across border of collarette two or three times and place in transport medium or sterile container |
|
Papule
|
Sampling by biopsy is probably more reliable. Provide local anesthesia by subcutaneous injection of 2% lidocaine. Clip hair with sterile scissors or clippers. Clean skin surface by a single wipe with 70% alcohol (no additional surgical preparation). Allow alcohol to dry. Using a sterile 3 or 4 mm punch and sterile surgical instruments, collect tissue sample and place in sterile container or transport medium. Suture biopsy site. Alternatively, papules may be prepared and disinfected as above, then sampled by insertion of a sterile needle and culture of emerging or expressed blood or exudate. |
For more information concerning canine pyoderma, please see the articles below.
- Guidelines for the diagnosis and antimicrobial therapy of canine superficial bacterial folliculitis (Antimicrobial Guidelines Working Group of the International Society for Companion Animal Infectious Diseases). Hillier A. et. al. Vet Dermatol. 2014 Jun; 25(3):163-75.
- Recognizing Pyoderma More Difficult than it May Seem. Gortel K. Vet Clin North Am Small Anim Pract. 2013 Jan; 43(1):1-18.
- Canine superficial bacterial folliculitis: Current understanding of its etiology, diagnosis and treatment. Bllom P. Vet J. 2014 Feb; 199(2): 217-222.
Dr. Ganta is an anatomic pathologist in the Kansas State Veterinary Diagnostic Laboratory and Dr. Bagladi-Swanson is a dermatologist in the Veterinary Health Center at Kansas State University.